 
Point to Remember: Mostly for transition and inner transition elements, the valence electrons are the electrons present in the shells outside the noble gas core. (See the above or below periodic table to see the valence electrons of inner transition metals). This is determined based on the number of electrons that would be added, lost, or shared if it reacts with other atoms. 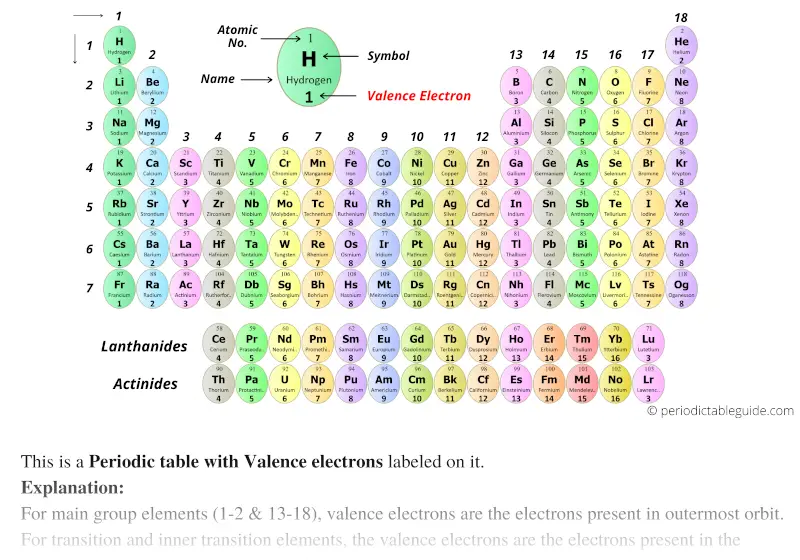
Valence describes how easily an atom or radical can combine with other chemical species. Inner transition elements can have valence electrons ranging from 3 to 16. 6: This version of the periodic table displays the electron affinity values (in kJ/mol) for selected elements. The words valence and valency have two related meanings in chemistry. In some inner transition metals, the electrons of incomplete d-orbitals are also considered as valence electrons. The periodic table shows the number of electrons in the valence shell of a chemical element that are available in the formation of a chemical bond. Hence, for inner transition elements, the electrons of both f-subshells as well as s-subshell behave like valence electrons. The inner transition elements have incomplete f- subshells and they are very close to the outer s-subshell. The number of valence electrons of an element can be determined by the periodic table group (vertical column) in which the element is categorized. (See the above or below periodic table to see the valence electrons of transition metals).Īlso the two bottom rows at the bottom of the periodic table are the inner transition elements (or f-block elements) also have the similar case. Hence, the transition elements (i.e d-block elements from group 3 to 12) can have more valence electrons ranging from 3 to 12. So, the electrons of both d-subshell and s-subshell behave like valence electrons. It is more difficult to find the valence electrons of transition elements as they have incompletely filled d-subshell and this d-subshell is very close to the outer s-subshell. What about valence electrons of transition and inner transition elements?įor the transition elements and inner transition elements, the case is more complicated. Well, this suits perfectly for the main group elements (i.e group 1, 2 and group 13 to 18), but what about the transition and inner transition elements? Hence, magnesium has 2 valence electrons. Here, you can see that the highest principal quantum number is 3, and the total electrons in this principal quantum number is 2. In this step, the electrons of cobalt have to be arranged. (b) Covalent radii of the elements are shown to scale. The atomic radius for the halogens increases down the group as n increases. Step-2: Need to do electron configuration of cobalt. 1: (a) The radius of an atom is defined as one-half the distance between the nuclei in a molecule consisting of two identical atoms joined by a covalent bond. That is, the cobalt atom has a total of twenty-seven electrons. From the periodic table, we see that the atomic number of cobalt is 27. The electron configuration of magnesium is 1s 2 2s 2 2p 6 3s 2. How do you calculate the number of valence electrons in a cobalt atom. Valence electrons can also be determined as the electrons present in the shell with highest principal quantum number (n). The magnesium element has 2 electrons in outermost orbit. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
